Reactivity trend across period 38/9/2023  Potassium reacts faster than sodium to produce hydrogen gas. Sodium reacts slowly with water to produce hydrogen gas. Click on the blue writing below to see the reactions. Nonmetal reactivity increases from left to right and decreases down groups. This page describes the reactions of the Period 3 elements from sodium to argon with water, oxygen and chlorine. (This is a greatly oversimplified explanation of acid-base chemistry.). nonmetallic characteristics increases from left to right and decreases down groups. Because the alkali metals are very reactive, they are seldom (if ever) found in. Our experts can deliver a Trends Across Period 3 Oxides and Chlorides essay tailored to your instructions for only 13.00 11.05/page. These metals react readily with water to produce hydrogen gas. Metal reactivity decreases from left to right across periods and increases down groups. In the same regard, the experiment was aimed at enabling the students to understand the trends in the chemical reactivity of these compounds and elements. AllĮlements in group 1 are very reactive metals, with the exception of hydrogen Can you explain the trend in atomic radius?Īll elements in a particular group share similar properties. The reactivity of metals decreases across the period from left to right, because metals still tend to get rid of more valence electron that needs a lot of. Look at the electronic configuration of each atom. 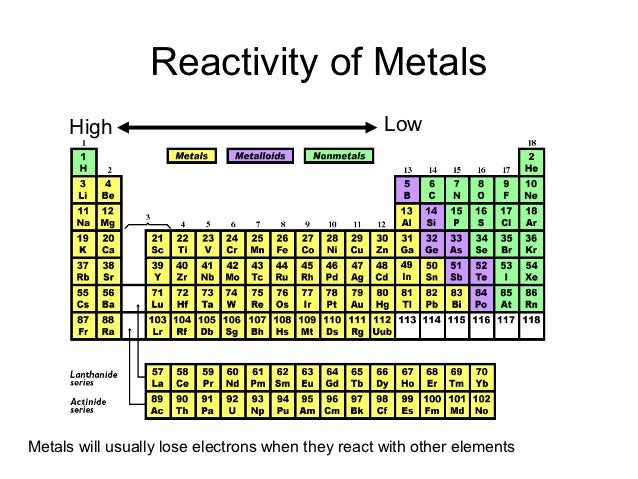
Nitrogen and phosphorus in group 15 usually form -3 ions. The alkaline earth metals always form +2 ions. Elements in the same group display similar chemical properties and trends immerge as we go along a period and down a group.Ĭlick to see how size of atoms changes across a period and down a group. ION CHARGE: - The number of electrons an element tends to gain or lose is a periodic property. The table is divided into groups(columns) and periods(rows). This is because since electrons are pulled closer to nucleus (see atomic radius), more. The modern periodic table has elements arranged in order of electronicĬonfiguration. 6 trends need to know: IE increases across period from left to right. Recognise that elements can be grouped together according to atomic mass. Mendelev was a Russian scientist who was first to The periodic table is a way of organizing known elements into groups with Phosphorus (P) has 5 valence electrons and needs 3. 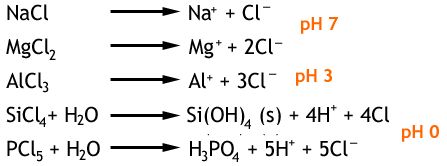 
Group 1 (Alkali Metals) are the most reactive compared to other metals. Moving further right in the period towards non-metals, the chemical reactivity again gradually increases. Content on this page requires a newer version of Adobe Flash Player. But, reactivity decreases when moving from left to right along a period. 
0 Comments
Leave a Reply.AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
